I'm on a quest around the world to find Bigfoot.
I'd originally set out to see if GlaxoSmithKline would actually help patients affected by Seroxat... but I decided to keep my goals realistic.
Here's why.
A short time ago I
highlighted the plight of "Zoe", a Seroxat sufferer who has been screaming for help in getting off her daily dose of Seroxat.
Zoe, you see, wants to try and kick her Seroxat addiction into touch. She's currently down to 2.4mgs and is having to use the sickly oral suspension that comes complete with an oral syringe.
She drops by 0.5mgs per week, anything higher is intolerable. It was myself who recommended to Zoe that 0.5mg would be slightly easier than a whole 1mg.
I've previously suffered the addictive qualities of Seroxat. It took me 18 to19 months to taper from 40mg per day to 22mg per day...in the end I went cold turkey. The 'two weeks' of tapering quoted by Alastair Benbow and Mary Anne Rhyne, both GlaxoSmithKline spokespersons, was poor advice based on...um...well, it's not known what their claims were based on. I have been asking GSK via the Freedom of Information Act but, despite two requests, they have failed to answer me. More about that later.
I gave Zoe the suggestion because neither her psychiatrist or doctor could recommend a tapering regime for her, neither could GlaxoSmithKline. Zoe had contacted them and asked for help... they told her to 'talk to her doctor.' When I say 'they' told her, I actually meant they got one of their highly paid lawyers to speak with her on the phone. Classy huh? Even more astonishing was this lawyer's apparent ability to predict the future. Zoe pressed him about the current UK Seroxat litigation, he replied by telling her there would be a major announcement about that soon. [Still waiting Mr Blabbermouth]
You see, there is no advice that GlaxoSmithKline can offer because they just don't know. The Seroxat clinical trials went on, at a guess, for approximately two years. Any person, today that goes over two years on Seroxat are basically venturing into the land of the unknown.
Another patient who contacted me [who wishes to remain anonymous] also wrote to GSK asking for help. Here's their reply:
 |
| Click on image to enlarge |
The frustrated patient wrote back an angry response to GSK:
I recently E mailed you concerning my terrible experience while discontinuing , and after Taking your so called wonder drug Seroxat !!
And I must say that I am not impressed with your response !! And that I refuse to sign the consent form to give you access to contact my GP and investigate my medical files !! why you may ask ?? I’ll tell you !! you only wish to investigate for your own benefit !! and have absolutely no intention or interest in helping myself or anyone else in my situation !! You manufacture these products so must have a resolution to put things right when they go wrong as they have !! DO YOU ?
The reply continued with...
Mr Witty and Mr Benbow should be thoroughly Ashamed of themselves for defending this Drug !! I hope you pass on this letter to them so they can read for themselves what SEROXAT has done to Me...
Benbow, not shy in front of TV camera's, accepts there may be patients who take longer to 'discontinue', Glaxo's spin for 'withdrawal'. He does not tell us why though, nor does he tell us how. Why? Because he doesn't know.
A university student also contacted me, he, just like the above two patients, was trying to taper from Seroxat. Things got so bad for this particular patient that even a reduction of 1ml per month on the liquid form of Seroxat was proving to be too difficult.
He was given a reduction plan by his local prescribed medication support service, a service ironically run by the NHS. He told me,
"I think part of the problem was making a reduction and then waiting for my brain to adjust. This method seems to attempt to get around that problem, and seems be set out over a realistic time span."
The current advice on the Seroxat patient information leaflet is to taper by 10mg per week. The above tapering plan appears to show that Glaxo's advice is way too ambitious - The above withdrawal plan, for the patient who is stuck on 10mg, is expected to last a staggering 71 weeks!
Don't you find it astonishing that the NHS acknowledge that SSRi withdrawal is severe, or at least this one small centre does, yet the manufacturers of these drugs and those that are supposed to regulate them, don't?
The Pwll Glas Resource Centre is a small centre servicing a small town [Mold] and the Prescribed Medicine Support Service is only a small part of what they do. The centre is mainly where Community Psychiatric Nurses [CPN's] are based and deal predominantly with mental health problems and run
Cognitive Behaviour Therapy counselling services. I wonder if they have contacted the manufacturers of these drugs and asked for help in getting their patients off them, I wonder if they were told to refer to the patient information leaflets? It's utterly absurd that we have an acknowledgement from a small centre in a small town which is run by the NHS, yet we have no acknowledgement from our own government, medicine regulators and pharmaceutical industry!
71 weeks is 1.3 years. An astonishing length of time to taper off a drug that Glaxo claim takes just 2 weeks!
I'm neither shocked or surprised at the attitude of GlaxoSmithKline with regard to the above three patients and for the hundreds of thousands who are guided by Glaxo's advice on the patient information leaflets. This isn't the first time they have shirked their responsibilities, it won't be the last.
In 2009 a jury concluded by a majority of 10-2 that Seroxat was the causation in Lyam Kilker being born with heart defects.
Internal documents to surface from that trial showed how Glaxo knew as early as 2001 that Seroxat may cause pregnant women to give birth to children with defects. The internal document showed how a woman had wrote to Glaxo in May 2001, she wrote:
**Paxil is the US brand name for Seroxat.
"I was diagnosed with panic disorder about four-and-a-half years ago. Since that time I've been taking Paxil, which is truly a miracle drug. I've been panic-free with this drug and have been able to go on with a normal life.
"I was married in October of 2000. My husband and I found out we were pregnant at Christmas time. I was so excited. I love children. The only problem is that I carried the baby to six months gestation and then had to have a termination.
"The doctors diagnosed my son with Truncus arteriosis. They said he would not lead a normal childhood and would most likely not make it through the open heart surgery that he would need as soon as he was delivered (if he was able to make it to that time).
"To say the least, I was absolutely distraught with this news. I thought this was something that I did [...] because I stayed on the Paxil for selfish reasons.
"I wanted to know if you could direct me to any information you might have of any woman that has taken Paxil and still had healthy babies.
"My husband and I are ready to try again to get pregnant in the next monthor two. I am so nervous. I don't want to stop taking my miracle pill. But, then again, if there is a chance that this might hurt or affect the baby, I want to know upfront. And I will somehow stop taking it for the time being.
"Please contact me as soon as possible. I love everything this drug has done for me. I am so thankful that your company had this available for me. I just want to continue to have a normal life and have the child that I always wanted.
"Please contact me as soon as possible ... Please don't forget about me."
The same woman sent a second email on June 1 2001, this time she wrote:
"This response is in regards to an e-mail that I had sent you previously. I was asking to see if you have any or are in the process of any clinical trials for women who are currently on Paxil and pregnant. I wanted to find out information to see how many women were on Paxil during pregnancy and if they were able to successfully have healthy babies.
"I am in no way insinuating your product did this to my child. I love the product, and I don't think I could have gotten through my panic attacks without the wonderful help of this miracle drug.
"I just want to start to try and get pregnant again soon. I do not want to put my unborn child through anything that would hurt him/her.
"Please, if you do not have this information, where is this information held? Does anyone do studies like this? Please, any information you may give me would be great. Thanks again for your help."
Glaxo responded to the distraught woman five days later on June 6 2001:
"Further questions about your treatment should be directed to the physician, pharmacist or healthcare provider who has the most complete information about your medical condition.
"Because patient care is individualised, we encourage patients to direct questions about their medical condition and treatment to their physician.
"We believe that because your physician knows your medical history, he or she is best suited to answer your questions.
"Our drug information department is available to answer any questions your physician or pharmacist may have about our products. Your healthcare professional can call our drug information department ..."
Once again, Glaxo were telling a patient to talk to their doctor!
What they didn't tell either this woman or her doctor was the discussions they had after they had received her emails.
An internal document shows them blaming their own drug Paxil [the US brand name for Seroxat]
Case number A0348482B", dated 13 June 2001, states: "Relatedness assessment to medication – almost certain."
Still, Glaxo never told this woman or her doctor! One has to ask the question why?
During testimony presented to a Pennsylvania jury, Glaxo officials indicated in 2001 that they were
“almost certain” that Paxil caused a heart defect in an unborn fetus. Former Glaxo drug-safety executive Jane Nieman testified that the determination was made by an unidentified Glaxo employee in the company’s database after the mother emailed the company inquiring if there was a connection between her use of Paxil and the birth defect.
In fact, Sean Tracey, the lawyer representing the Kilker family, said in his opening statement of the trial that Glaxo executive, Bonnie Rossello, wrote in a 1997 memo on what the company would do if forced to conduct animal studies on the drug. She wrote,
“If neg results. can bury.” This statement by Rossello was to do with withdrawal.
Do you see a pattern forming here?
Janice Simmons, who operates the
Seroxat User Group, has amassed over 60,000 emails from Seroxat patients, most of them are struggling to get off Seroxat. Janice cannot tell them how to combat the side effects such as; electric zaps, suicidal thoughts, intolerance to sudden loud noises, bouts of crying, headaches and visionary disturbances etc.
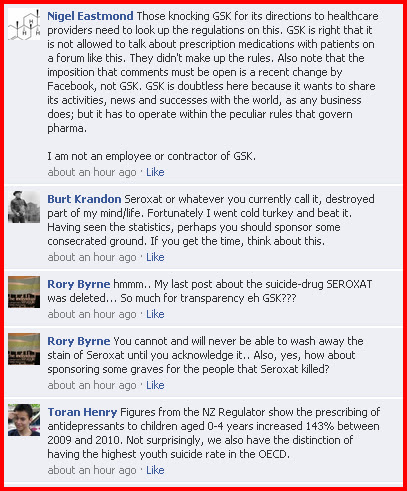
So, Janice decided to request a meeting with GlaxoSmithKline's CEO, Andrew Witty. To her surprise, GSK’s UK medical director Dr Pim Kon wrote back asking what she wished to discuss...adding that they cannot give advice to patients, they need to seek that from their healthcare professionals. [Yawn]
Janice informed Dr Kon that she wished to discuss the issue of Seroxat withdrawal. Kon wrote back telling Janice that they was not allowed to discuss personal matters with patients and that they should
'talk to their doctor'. [Another yawn]
Is that building in Brent surrounded by mountains? The echo is unmistakably obvious.
YODEL-AY-EE-OOOO
Kon added that the Seroxat patient information leaflet had been updated with information about [ahem] 'discontinuation' .
Dr Pim Kon is no stranger to controversy. Last year she told the
InPharma magazine that European regulators [EMA] were wrong to withdraw its diabetes drug Avandia from the market. Avandia was suspended by the EMA last year because of high levels of heart attack, heart failure and strokes in patients
Let's take a gander at
Glaxo's Seroxat withdrawal advice:
Do not stop taking Seroxat until your doctor tells you to.
When stopping Seroxat, your doctor will help you to reduce your dose slowly over a number of weeks or months - this should help reduce the chance of withdrawal effects. One way of doing this is to gradually reduce the dose of Seroxat you take by 10 mg a week.
10 mg per week? Ah, I can see why they have plucked this amount out of thin air. The maximum dosage for mild depression is 20mg, if you drop by 10mg per week Glaxo have their
"It takes the majority of patients two weeks to discontinue." I'm reminded of a song by Ian Dury and the Blockheads, "There Ain't Half Been Some Clever Bastards" - (Dury, Jankel - 1979)
INTERLUDE
Janice wrote back to Kon and asked if GSK could provide her with any studies that GSK had carried out on withdrawal from Seroxat to back up GSK's statement that 'it may be helpful to reduce by 10mg per week and withdraw over the course of 2 weeks. - Kon, after a period of two weeks, has failed to provide Janice with any such evidence.
So, it's a given that I am not fond of GlaxoSmithKline, in particular their spokespersons Mary Anne Rhyne and Alastair Benbow. I'm not particularly fond of their highly paid lawyers either, have any of them ever seen the movie
The Devil's Advocate? Keep on telling yourselves that you are doing a great job folks. You know, I'd really like to see things from your point of view but I can't seem to get my head that far up your arses!.
Here's the paradox. GSK refuse to help patients who contact them with regard to difficulties weaning off Seroxat. They quote the ABPI Code of Practice and tell the patient to 'talk to their doctor'. The doctor either thinks their patient is still suffering from the original illness or knows that the drug is addictive but still prescribes it anyway!
A recent survey showed that almost 80% of doctors routinely prescribe drugs to which they believe the patient may be addicted! GSK's lawyers support them [GSK] to the hilt despite witnessing the devastation this drug has caused. Round and round we all go, caught in the psychopharmaceutical trap folks. It's a merry-go-round that isn't very merry at all. We want to get off but the pharmaceutical industry, doctors, psychiatrists and lawyers won't let us, they just keep on spinning the ride and the truth.
By Glaxo's own admission, Seroxat can cause withdrawal problems for some. Yet they have defended every single accusation that Seroxat has caused problems for their consumers, instead they blame the original illness or other mitigating factors, anything but the very same drug they say 'can' cause withdrawal problems for some people. Glaxo's Seroxat now carries a warning for pregnant mothers, yet every single mother whose child has been born with defects get no admission of liability from Glaxo. Glaxo's wonder pill can also cause suicide, once again they have never once admitted liability for any suicide on Seroxat. They defend this medication yet say all of the above are possible. That's like a chef presenting you with a steak, the steak may kill you or give you botulism but the Chef, although admitting that the steak he cooked could be harmful, will deny any wrongdoing if you pursue the matter, he, in actual fact, will blame everything...but the steak. It's GSK having their cake and eating it.
The Olive Branch snapped in half.
Had Witty been brave enough to meet with Janice Simmons I would have just loved to have been present. I would have liked him to help people come to terms with their Seroxat addiction, I would have liked him to acknowledge that people, like Zoe, need help and I would have liked his company to pay for that help by way of offering her, and people like her, a placement on a detox program.
I would have also have liked Andrew Witty to help those who have been off Seroxat for sometime but have been left with long term side effects. Long term effects that they, once again, deny.
Now, Witty, and indeed all of Glaxo's spokespersons, can continue down the road they've all been stuck on for all these years and deny Seroxat causes a myriad of side effects or they can start helping those that their product has harmed.
Any response along the lines of,
"Seroxat is not addictive" or
"Seroxat has benefited millions of people worldwide" would have been seen as an attempt of impunity and would have been greeted with a two finger salute, by me at least. It would also have been acknowledged that the only reason GSK roll out these boring statements is to protect themselves from litigation.
On the subject of litigation, I would have liked Andrew Witty to give those that have been harmed by his company's drug some closure. Let them be allowed to turn around to all those finger pointers and say,
"I told you it was a side effect and not the 'real' me."
I guess any admittance would have had repercussions on the benefits system here in the UK too. Any acknowledgement that Seroxat can debilitate people during withdrawal and even years after stopping it could see a deluge of disability claims. The government wouldn't want that now, would they? And with Mr Witty appointed as Lead Non-Executive Board Member for the Department for Business, Innovation and Skills
in 2010, I can only assume that they would take a dim view if he were to admit his company's product harmed people to the point where they could not function properly. I can only begin to imagine the amount of money Mr Witty is saving the UK government by not announcing that his company's product has caused so much pain and misery to thousands of UK residents. Of course, he will argue that it has helped
"millions of people worldwide", which doesn't really address the outstanding issue, does it?
You see Mr Witty, I've got the staying power of an energy saving light bulb. I can, and will continue to highlight articles in the press regarding your company. I'll also continue to offer readers of this blog court documents if and when they become available, documents from the USA where your attorneys have struck deals with claimants, only to be silenced by gagging orders because, it would appear, you want the truth suppressed.
I'm like one of those little bunnies that play the drums in the TV adverts, I can keep on going because I've been loaded with the Duracell batteries and not the normal ones, capice?
Oh, and please don't quote the safety reviews carried out by the 'expert' working committees either. An expert would be able to tell me and the thousands of others how to wean off your product without having to experience the intolerable side effects for days, weeks, months and years on end. The committees are basically the MHRA with a few 'outsiders' thrown in to show there is no conflict of interest...we all know there is though.
Any association with the MHRA is seen, by me at least, as a total embarrassment. Both the expert working group and MHRA are clueless and, it would appear, they have no desire to change. You could, however recommend a decent shampoo for employees of the MHRA, their scalps must be terribly itchy with all that sand their heads have been buried in all these years. Not to mention the kind of 'expert' witnesses Glaxo's lawyers have called upon to defend their product in the current UK Seroxat litgation. Remind me who
Rashmi Shah used to work for again please?
- Dr. Rashmi Shah is the owner of Rashmi Shah Consultancy Ltd, located in Slough, Berkshire, UK.
- Shah was employed by the MHRA between 1987 and 2004. Positions held were: Senior Medical Officer, Senior Clinical Assessor and Senior Medical Assessor.
- An employee of the MHRA for 17 years.
The regulator and Pharma, as one. Snug as a bug on a rug.
Let's not forget the head of licensing at the MHRA, Dr Ian Hudson, whom were his previous employers?
SmithKline Beecham!
And his role at SmithKline Beecham?
World Safety Officer!
In fact, one of Hudson's significant involvements was with paroxetine, that's Seroxat for those that don't know. [
Source]
Is it any wonder the MHRA give Seroxat a clean bill of health each time it is reviewed?
Their Chairman's embarrassing performance in front of TV camera's trying to defend Seroxat was plain to see for the millions that tuned in BBC's
Panorama exposé. And who is their Chairman?
Alasdair Breckenridge.
His former employers?
GlaxoSmithKline [Formerly SmithKline Beecham]
To coin a phrase, "This crap just writes itself."
You want to improve Glaxo's image? Make a stand, be the first pharmaceutical company to hold up their hands and say,
"You know what, the customers are right...they do need our help." I guess that would boil down to an admission of guilt though. When was the last time Glaxo ever admitted any wrong-doing...apart from when they were forced to?
An apology would have been nice but I can't say for sure if it would have been accepted by the likes of the
Gatchell,
Carlin and
Keegan families, whom have each lost a precious life because they actually believed it when they were told their teenage children would be okay taking your product.
It takes a big man to step up to the plate, are you that man Mr Witty? Obviously not.
Can you imagine how good it would have felt if you, Mr Witty, were part of the solution?
Alas, it appears you are not up to the challenge. Why am I not surprised! Maybe you are allergic to people? Isn't that how Glaxo first promoted Seroxat in the US with a huge poster campaign? [
SOURCE]
Seroxat is rarely prescribed any more in the UK, recent figures show that it is, in fact, the least prescribed SSRi in the UK today - why is that?
Don't bother answering...we all know why, don't we?
I am left wondering how your company came up with the idea to treat depression, anxiety and shyness. Did you ever think about how one could be diagnosed? It amazes me that psychiatrists and doctor's can diagnose a patient without any form of removal of bodily fluids. Nothing was taken from my brain when I was prescribed Seroxat nor did I have to urinate into a jar or have blood taken to show I had the blues.
One would think that a manufacturer of a product would act upon a defective product, particularly if that product was to be used by millions of people. Then again, sadly, the UK public aren't even considered when it comes to your company acknowledging Seroxat withdrawal. Readers of this blog will be surprised to know that GlaxoSmithKline have made over 3,000 out-of-court settlements in America, all those settlements were to do with the withdrawal issue. A settlement means no liability, once again Glaxo can refer those left suffering to their doctors.
Glaxo launched Seroxat in direct competition to Eli Lilly's Prozac. Lilly, being Lilly, saw this as a threat. So battle did commence.
According to this recently surfaced internal file, GlaxoSmithKline [then SmithKline Beecham] knew of two studies that Lilly had carried out, those studies showed that Seroxat showed significantly higher rates of withdrawal problems when compared to Prozac.
How did GSK handle this?
Well, instead of addressing Lilly's findings they decided to play down the issue of Seroxat withdrawal. The fact that Seroxat withdrawal problems were significantly higher than Prozac saw Glaxo's team pull together the following plan.
 |
| Click to enlarge |
 |
| Click to enlarge |
You will note the date of the memo is July 1997, that's 14 years ago!
It would appear, Mr Witty, that your company were already debunking the withdrawal problem with spin way back then.
Personally, I believe your company royally ballsed up by not carrying out any extensive studies into the protracted withdrawal of your product, choosing instead to set money aside for future out of court settlements to consumers that had to go through the legal system to get some kind of justice after being harmed by your drug. Money well spent for Glaxo, probably all part of the insurance cover your company takes out when they launch a product. I believe that money would have been better used on a withdrawal study, then again you already knew about the withdrawal problems early on, didn't you...well maybe you didn't but your predecessor, JP Garnier,
must have known, right?
Is it any wonder that you have decided not to manufacture or invest any more of your billions into antidepressant medication?
"You don't need a weatherman to know which way the wind blows" -
Bob Dylan
I'd also be grateful if you could ask your company's spokespersons to put a lid on making statements every time another child or teenager dies whilst on your product. Statements with the tired out phrases,
"we utterly refute" and
"Seroxat has benefited millions of people worldwide" really do nothing to ease the pain of the parents, brothers and sisters left behind.
It would be real nice if you could chase up a couple of Freedom of Information requests, they are just basically questions, I understand that, even by using the FOIA, you are not obliged to answer... be worth your while if you did though. I will understand if you don't want to - any answers would kind of leave a door open to further probing of Seroxat, wouldn't it?
GSK Customer Relations - UK
The following request I make under the Freedom of Information Act.
To whom it may concern,
1. In a 2002 interview with BBC TV's Shelley Jofre, GlaxoSmithKline spokesperson, Alastair Benbow, claimed that, "... the majority of patients who experience withdrawal symptoms - and the majority of patients actually do not experience any withdrawal symptoms - of those that do the majority of those symptoms are mild to moderate in nature and will go away without any treatment within two weeks." He was, of course, referring to the antidepressant Seroxat. Under the FOIA I would like GlaxoSmithKline to provide me with the study/studies that substantiate Dr Benbow's claim.
2. Furthermore, in the same interview, Dr Benbow claimed, "maybe that a small proportion of patients do get more severe symptoms...". Under the FOIA I would like GlaxoSmithKline to provide me with the study/studies that substantiate Dr Benbow's claim.
The full transcript, which has recently been released, was part of disclosure and used in litigation against GSK in the United States and is attached for your perusal.
Yours sincerely,
Bob Fiddaman
And this one to GSK Australia:
I've recently learned that GSK Australia rolled out the a plus project in 2002 The project, as I understand, was for the promotion of the antidepressant Aropax.
My questions, submitted to you under the FOIA, are thus:
1. How many patients were enrolled in the a plus project?
2. How many of the patients were under the age of 18?
3. Did the starter packs just contain packets of Aropax with 20mg tablets or were there starter packs with lower doses?
4. What were the qualifications and training of the counsellors on the a plus project?
5. What were the sales trends of Aropax in Australia before, during and after the a plus project?
6.How much did GlaxoSmithKline pay individual key opinion leaders, including (but not limited to) Professor Graham Burrows and Professor Bruce Singh? What were the services for which each was paid?
7. What were the goals of the a plus project?
8. How long did the a plus project run for?
9. How many adverse reactions were reported for Aropax during the project?
10. Were participants on the a plus project ever followed up or did GlaxoSmithKline Australia lose touch with the participants once the project had finished?
11. Why was the project ended?
12. Was the a plus project deemed a success by GlaxoSmithKline?.
13. Has GlaxoSmithKline or its subsidiaries ever offered similar counselling sessions to patients taking other drugs manufactured by the company?
14. How much did GlaxoSmithKline pay for sponsorship of the Depression Awareness Journal around the time of the a plus project?
Mr Bob Fiddaman
Chase these FOI requests for me Mr Witty, there's a good chap.
Finally, could you ask GSK Japan to justify the money they have recently spent on a Seroxat clinical trial aimed at
7-17 year old children.
Oh, don't bother screaming like a big girl to your lawyers. I'm merely asking for things and making a mockery of your company and their stance toward making people
do more, feel better and live longer. Ironic that you have never changed that motto despite all those that have been harmed by your drugs.
I'm ashamed that GlaxoSmithKline are a British company. I'm astounded that you continue to refuse to help patients who are suffering at the hands of your product. To me that's an intention to inflict emotional distress, you merely hide behind the Association of the British Pharmaceutical Industry code of practice, a great weapon of mass deflection you have there Mr Witty. Ooh look I just did a play on words,
"Weapon of mass deflection." [Scroll up and play Ian Dury again if you feel inclined]
Our current government, in fact any party voted in, see you as the jewel in the crown.
One only has to look at the 2001 Pharmaceutical Industry Competitiveness Task Force Final Report to see how the industry is revered.
Page 20 - 'The UK benefits in the following ways from the pharmaceutical industry...
Returns to UK shareholders, tax revenues accruing to the UK Exchequer and benefits arising from terms of trade.
I can only guess that if they were to ever mess with GSK, you could always move your business elsewhere. That's quite a grip of the gonads Glaxo have on the UK economy.
Looks like I have chosen my unpaid career as someone who will continue to write about you and your company until you put things right. Any threatening letters by your lawyers will be copied and re-published on this blog.
I'd copy in the press with this post but seeing as you have one of the Murdoch's on your board at GSK I can only assume that they will only print what you want them to. Remind me, what exactly is James Murdoch's role at GSK? Did he have any input into your promotion/protection of the MMR Jabs? There are many that are suggesting he did. [
Rupert Murdoch, Brian Deer, Dr Andrew Wakefield and fabricated lies]
Tut tut - children the targets yet again!
I guess it will all come out in the wash eh Mr Witty? - Just like your product, Ribena, you know, the one that you said was "Full of Vitamin C", when actually it wasn't! [
SOURCE]
It's entirely up to you whether you wish to reply, I doubt you will.
**Inserts Duracell battery and proceeds to bang the drum.
Bob Fiddaman
Former Seroxat addict
Left with long term adverse reactions due to ingesting Seroxat for 6 years
"When stopping Seroxat, your doctor will help you to reduce your dose slowly over a number of weeks or months - this should help reduce the chance of withdrawal effects. One way of doing this is to gradually reduce the dose of Seroxat you take by 10 mg a week." -
Seroxat Patient Information Leaflet 2010